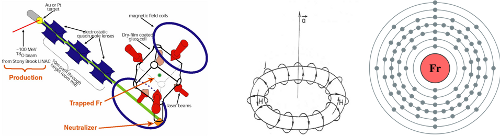
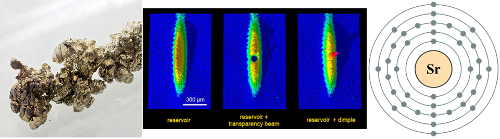
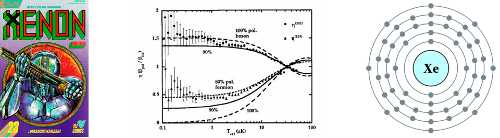
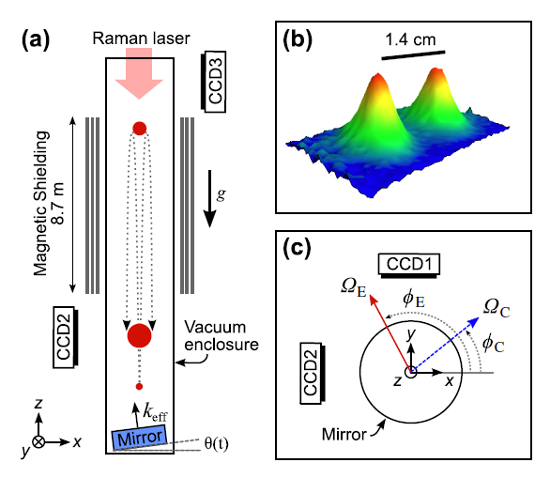
Element: Lithium (Li) Atomic Number: 3 Mass: Two stable isotopes, masses 6 and 7 amu Laser cooling wavelength: 671 nm Doppler cooling limit: 140 μK. Chemical classification: Alkali metal, column I in the periodic table. Yet another greyish metal. We’re almost done with alkalis, I promise. Less reactive than any of the others, so the […]